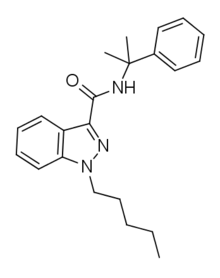
CUMYL-PINACA powder designer cannabinoid and a great analogue of marijuana. It has long established itself in the market for research chemicals, as a cannabinoid. Therefore, true appreciated qualities continue to conduct their experiments with this cannabinoid.



















Reviews
There are no reviews yet.